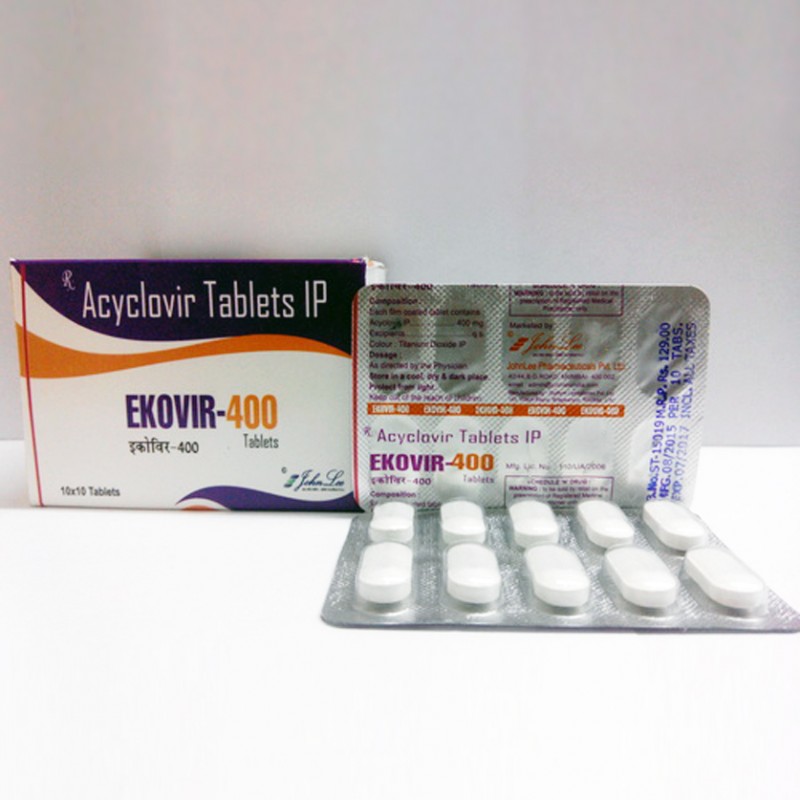
Ekovir John Lee
$4.40
400mg (5 pills)John Lee
Description
Product Description
Aciclovir (ACV), also known as acyclovir, is an antiviral medication. It is primarily used for the treatment of herpes simplex virus infections, chickenpox, and shingles. Other uses include prevention of cytomegalovirus infections following transplant and infections due to Epstein-Barr virus. It is available by mouth and intravenously.
Common side effects include nausea and diarrhea. Potentially serious side effects include kidney problems and low platelets. Greater care is recommended in those with poor liver or kidney function. It is generally considered safe for use in pregnancy with no harm having been observed. It appears to be safe during breastfeeding. Aciclovir is a nucleic acid analogue made from guanosine. It works by decreasing the production of the virus’s DNA.
The discovery of aciclovir was announced in 1977. It is on the World Health Organization’s List of Essential Medicines, the most important medications needed in a basic health system. It is available as a generic medication and is marketed under many brand names worldwide. The wholesale cost as of 2014 to 2016 was between US$0.03 and US$0.12 for a typical dose by mouth. The cost of a typical course of treatment in the United States is less than US$25.
Medical use
400 mg pills of aciclovir
Aciclovir is used for the treatment of herpes simplex virus and varicella zoster virus infections, including:
Genital herpes simplex (treatment and prevention)
Neonatal herpes simplex
Herpes simplex labialis (cold sores)
Shingles
Acute chickenpox in immunocompromised patients
Herpes simplex encephalitis
Acute mucocutaneous HSV infections in immunocompromised patients
Herpes of the eye and herpes simplex blepharitis (a chronic (long-term) form of herpes eye infection)
Prevention of herpes viruses in immunocompromised people (such as people undergoing cancer chemotherapy)
Oral aciclovir, however, does not appear to decrease the risk of pain after shingles. In those with herpes of the eye, aciclovir was found to be more effective than idoxuridine or vidarabine in relative number of successfully healed eyes.
Intravenous aciclovir is effective to treat severe medical conditions caused by different species of herpes virus family includes severe localized infections of herpes virus, severe genital herpes, chickenpox & encephalitis. It is also effective in systemic or traumatic herpes infections, eczema herpeticum and Herpes simplex meningitis. Reviews of research dating from the 1980s show there is some effect in reducing the number and duration of lesions if aciclovir is applied at an early stage of an outbreak. Recent research shows effectiveness of topical Aciclovir in both the early and late stages of the outbreak as well as improving methodologically and in terms of statistical certainty from previous studies. Aciclovir trials show that this agent has no role in preventing HIV transmission, but it can help slow HIV disease progression in people not taking anti-retroviral therapy (ART). This finding emphasizes the importance of testing simple, inexpensive non-ART strategies, such as aciclovir and cotrimoxazole, in people with HIV.
Pregnancy
Classified as a Category B Drug, the CDC and others have declared that during severe recurrent or first episodes of genital herpes, aciclovir may be used.[ For severe HSV infections (especially disseminated HSV), IV aciclovir may also be used. Studies in mice, rabbits and rats (with doses more than 10 times the equivalent of that used in humans) given during organogenesis have failed to demonstrate birth defects. Studies in rats in which they were given the equivalent to 63 times the standard steady-state humans concentrations of the drug on day 10 of gestation showed head and tail anomalies.
Aciclovir is recommended by the CDC for treatment of Varicella during pregnancy, especially during the second and third trimesters
Aciclovir is excreted in the breast milk, therefore it is recommended that caution should be used in breast-feeding women. It has been shown in limited studies that the nursing infant is exposed to approximately 0.3 mg/kg/day following oral administration of aciclovir to the mother. If nursing mothers have herpetic lesions near or on the breast, breast-feeding should be avoided.
Adverse effects
Systemic therapy
Common adverse drug reactions (?1% of patients) associated with systemic aciclovir therapy (oral or IV) include: nausea, vomiting, diarrhea, encephalopathy (with IV use only), injection site reactions (with IV use only) and headache. In high doses, hallucinations have been reported. Infrequent adverse effects (0.1–1% of patients) include: agitation, vertigo, confusion, dizziness, oedema, arthralgia, sore throat, constipation, abdominal pain, hair loss, rash and weakness. Rare adverse effects (<0.1% of patients) include: coma, seizures, neutropenia, leukopenia, crystalluria, anorexia, fatigue, hepatitis, Stevens–Johnson syndrome, toxic epidermal necrolysis, thrombotic thrombocytopenic purpura and anaphylaxis. Intravenous aciclovir may cause reversible nephrotoxicity in up to 5% to 10% of patients because of precipitation of aciclovir crystals in the kidney. Aciclovir crystalline nephropathy is more common when aciclovir is given as a rapid infusion and in patients with dehydration and preexisting renal impairment. Adequate hydration, a slower rate of infusion, and dosing based on renal function may reduce this risk.